CPT Infrastructure:
CPT includes the following subjects:
Institute of Physiology CAS, Department of Biological Control: test facility, GLP certified, non-clinical safety testing on rodents (mouse, rat, guinea pig) and non-rodents (rabbit)
[ezcol_1quarter]
 [/ezcol_1quarter]
[ezcol_1quarter]
[/ezcol_1quarter]
[ezcol_1quarter] [/ezcol_1quarter]
[ezcol_1quarter]
[/ezcol_1quarter]
[ezcol_1quarter] [/ezcol_1quarter]
[ezcol_1quarter_end]
[/ezcol_1quarter]
[ezcol_1quarter_end] [/ezcol_1quarter_end]
[/ezcol_1quarter_end]
Institute of Physiology CAS, Department of Metabolomics: test facility, GLP certified, chemical analysis of potential drug candidates and approved drugs in biological materials using liquid chromatography–mass spectrometry (LC–MS). The department also provides analysis of complex lipids (lipidomics), polar metabolites (metabolomics) and various exposome compounds (exposomics)

Institute of Molecular Genetics CAS, Czech Centre for Phenogenomics : test site, established quality system, histopathology, clinical chemistry, hematology, imaging, immunology, neurophysiology, cardiology, PoC pharmacology on xenografts;
[ezcol_1fifth] [/ezcol_1fifth]
[ezcol_1fifth]
[/ezcol_1fifth]
[ezcol_1fifth] [/ezcol_1fifth]
[ezcol_1fifth]
[/ezcol_1fifth]
[ezcol_1fifth] [/ezcol_1fifth]
[ezcol_1fifth]
[/ezcol_1fifth]
[ezcol_1fifth] [/ezcol_1fifth]
[ezcol_1fifth_end]
[/ezcol_1fifth]
[ezcol_1fifth_end] [/ezcol_1fifth_end]
[/ezcol_1fifth_end]
Institute of Animal Physiology and Genetics CAS, Centre Pigmod: test site, established quality system, non-clinical safety testing on non-rodents – minipigs, biocompatibility studies, ADME, animal models
[ezcol_1fifth]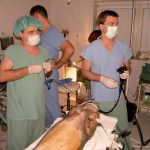 [/ezcol_1fifth]
[ezcol_1fifth]
[/ezcol_1fifth]
[ezcol_1fifth] [/ezcol_1fifth]
[ezcol_1fifth]
[/ezcol_1fifth]
[ezcol_1fifth] [/ezcol_1fifth]
[ezcol_1fifth]
[/ezcol_1fifth]
[ezcol_1fifth]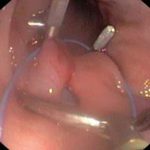 [/ezcol_1fifth]
[ezcol_1fifth_end]
[/ezcol_1fifth]
[ezcol_1fifth_end] [/ezcol_1fifth_end]
[/ezcol_1fifth_end]
Institute of Biotechnology CAS, Service Technological Laboratory: custom chemical synthesis of promising therapeutic substances, development of application formulations)

